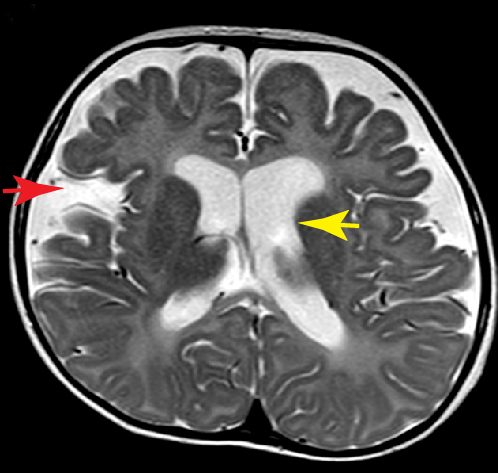
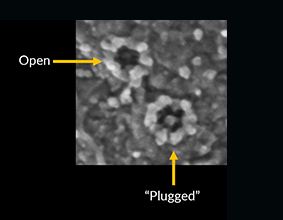
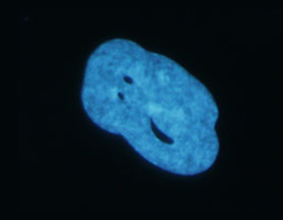
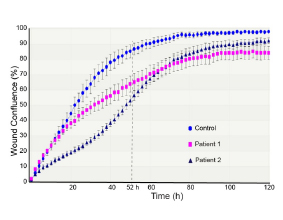
Nuclear envelopathies are a heterogeneous group of human diseases caused by mutations in genes encoding nuclear envelope and NPC proteins. Two of our recent research publications (Fichtman et al., American Journal of Human Genetics and Nature Communications, 2019) describe novel diseases from this group, involving drastic damage to the central nervous system. In the case of Nup214, a protein residing on the cytoplasmic face of the NPC, mutations cause severe neurodevelopmental regression, progressive microcephaly and cerebellar atrophy. SEM imaging showed a large increase in the presence of central particles (or ‘‘plugs’’) in the NPC channels of patient cells, suggesting that large transport cargoes are delayed in passage through the NPC. This is the first time that direct imaging at the single NPC level has revealed a disease-associated phenotype in human nuclear pores. A different disease is caused by the combined loss of protein isoforms LAP1B and LAP1C from the nuclear envelope. In this case, a devastating disease with severe neurological impairment and multiple effects in other tissues is detectible at birth. Our studies in patient fibroblasts revealed changes in the nuclear envelope and lamina, peculiar cytoplasmic channels cutting across the nucleus and a strong inhibition of cellular motility. The continued study of these diseases is now leading us into work with the hardest hit targets in the human body: neuronal cells. In collaboration with Achia Urbach’s lab, we are generating patient-specific induced pluripotent stem cells (iPSCs) and differentiating these into neurons and other cell type-specific models. In analyzing the effects of specific genetic mutations in this system, we are concentrating on nuclear envelope-induced defects in cell motility as well as the activation of various cellular stress response pathways. Other emerging connections between the nucleocytoplasmic transport machinery and neurodegeneration are reviewed in Fahrenkrog & Harel (Cells, 2018).

Nuclear Envelopathies 
Open & Plugged Human NPCs 
Distorted LAP1 Nucleus 
Cell Motility Assay 
Cell Motility Assay